
Clinical Development
Clinical Trials
Alcohol-Associated Hepatitis (AHFIRM)

We have completed a Phase 2b study (AHFIRM) in subjects with Alcohol-associated Hepatitis to evaluate saFety and effIcacy of LaRsucosterol treatMent (NCT04563026).
AHFIRM was a randomized, double-blind, placebo-controlled trial conducted at more than 60 clinical trial sites across the U.S., EU, U.K., and Australia.
The outcomes measured were 90-day mortality or liver transplant, 90-day mortality, 28-day mortality or liver transplant, and 28-day mortality in patients treated with larsucosterol plus standard of care compared to those treated with placebo (standard of care with or without steroids).
Expanded Access Policy
After careful review, DURECT has determined that it cannot offer its product candidates for expanded access (sometimes called “compassionate use”) at this time. DURECT believes that investigational products should be studied in patients as part of clinical trials designed to obtain data on safety and efficacy.
If you have questions about DURECT’s expanded access policy, please email us at medaffairs@DURECT.com.
Pipeline
Preclinical
Phase 1
Phase 2
Phase 3
Approved
Alcohol-Associated Hepatitis (AH)
Intraveneous administration
Preclinical
Phase 1
Phase 2
Phase 3
Marketed
Hematology/Oncology
Preclinical
Phase 1
Phase 2
Phase 3
Marketed
Larsucosterol has demonstrated promising results in human studies, including a Phase 2b trial in AH patients (AHFIRM), a previous Phase 2a trial in AH, and an open label Phase 1b trial in metabolic dysfunction-associated steatohepatitis (MASH).
Given the clinical profile to date, we believe larsucosterol has the potential to treat multiple serious and life-threatening conditions, including acute organ injury and cancer.
Preclinical
Phase 1-3
Marketed
(Non-opioid bupivacaine solution)
Post surgical pain (following arthroscopic subacromial decompression)
Preclinical
Phase 1-3
Marketed
POSIMIR® is sold in the U.S. by Innocoll Pharmaceuticals
DURECT maintains ex-U.S. rights
Alcohol-Associated Hepatitis (AH)
AH is a life-threatening form of alcohol-associated liver disease (ALD), which can occur in individuals who chronically misuse alcohol – frequently after increased consumption – regardless of age, gender, education or income. It is characterized by severe inflammation and destruction of liver tissue (i.e., necrosis). AH accounted for roughly 164,000 hospital admissions in the U.S. in 2021.1 Approximately 30% of patients die within 90 days of hospitalization.2 The charges for each hospital admission that resulted in death averaged approximately $180,000.1 There is currently no FDA or EMA approved treatment for AH, and novel therapeutic strategies are needed to improve survival.3,4 A number of clinical practice guidelines, including from AASLD and ACG, recommend corticosteroids as first-line treatment for severe AH despite limited and inconsistent survival benefits and widely acknowledged contraindications.5,6,7 Although liver transplantation is indicated for patients with severe AH, the procedure is costly ($875,000 per patient on average8), requires lifelong immunosuppression, and is limited by the availability of donated organs.6,9 Despite these drawbacks, the rate of liver transplantation for AH has increased rapidly in the U.S.10
Epigenetic modulation in AH
Larsucosterol potential in AH
In a Phase 2b trial (AHFIRM) to evaluate larsucosterol’s safety, efficacy, and potential lifesaving capacity for the treatment of patients with severe AH, larsucosterol data showed compelling mortality reduction at 90 days, compared to placebo. Topline results included:
- A pronounced reduction in mortality at 90 days in the U.S. population:
- A 57% reduction with the 30 mg dose of larsucosterol
- A 58% reduction with the 90 mg dose of larsucosterol
- A compelling trend in the key secondary endpoint of mortality reduction at 90 days in the global population:
- A 41% reduction with the 30 mg dose of larsucosterol
- A 35% reduction with the 90 mg dose of larsucosterol
- The numerical improvement in the primary endpoint of mortality or transplant at 90 days did not achieve statistical significance
- Safety: Both doses of larsucosterol were well-tolerated
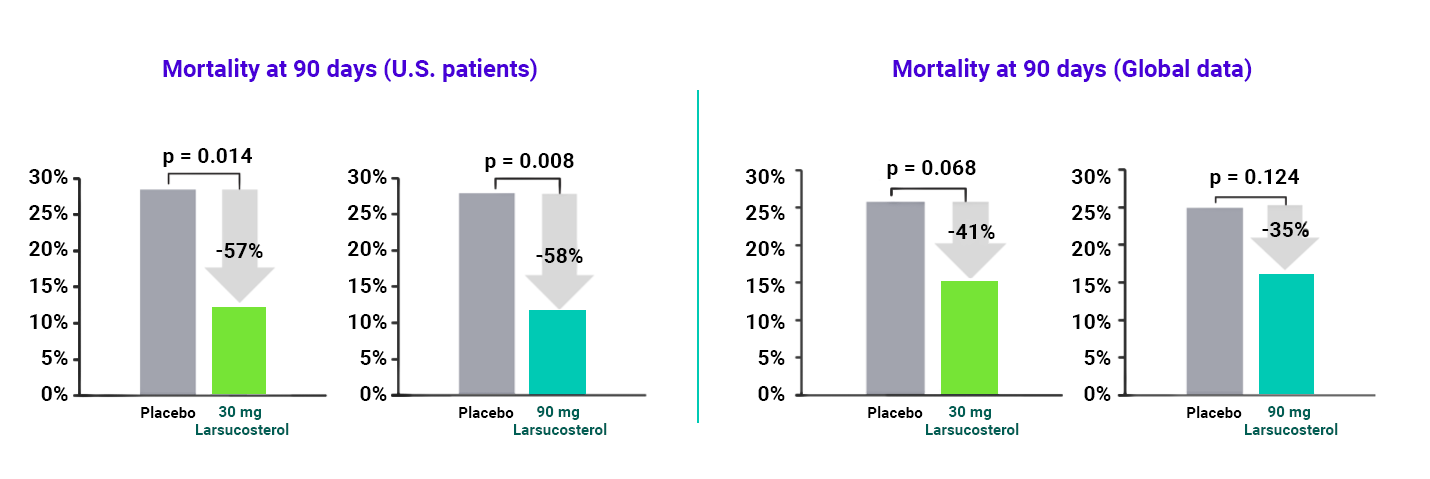
AHFIRM was a randomized, double-blind, placebo-controlled, international study, comprised of three arms with 307 total patients (approximately 100 patients per arm): (1) Placebo, which consisted of standard of care with or without steroids, (2) larsucosterol (30 mg), and (3) larsucosterol (90 mg).
Additional Larsucosterol Studies
The potential of larsucosterol has been evaluated in human studies beyond AH. Larsucosterol was evaluated in a Phase 1b clinical trial in patients with metabolic dysfunction-associated steatohepatitis (MASH) with stage 1 to 3 fibrosis, showing signals of positive data with no drug related serious adverse events to date.13 Results included improved liver enzymes, serum lipid profiles and certain biomarkers, and liver fat content. It showed improvement from baseline in insulin resistance and liver stiffness. Larsucosterol was well tolerated in all subjects.
References
- Data on File
- Hughes E, Hopkins LJ, Parker R. Correction: Survival from alcoholic hepatitis has not improved over time. PLoS One. 2018;13:e0195857.
- Thanda Han MA, Pyrsopoulos N. Emerging therapies for alcoholic hepatitis. Clin Liver Dis. 2021;25:603-624.
- Vergis N, Atkinson SR, Thursz MR. The future of therapy for alcoholic hepatitis – Beyond corticosteroids. J Hepatol. 2019;70:785-787.
- Crabb DW, Im GY, Szabo G, Mellinger JL, Lucey MR. Diagnosis and treatment of alcohol-associated liver diseases: 2019 practice guidance from the American Association for the Study of Liver Diseases. Hepatology. 2020;71:306-333.
- Singal AK, Bataller R, Ahn J, Kamath PS, Shah VH. ACG clinical guideline: alcoholic liver disease. Am J Gastroenterol. 2018;113:175-194.
- Shipley LC, Singal AK. Liver transplantation for alcoholic hepatitis. Transl Gastroenterol Hepatol. 2020;5:26.
- Bentley TS, Ortner NJ. 2020 U.S. organ and tissue transplant: cost estimates, discussion, and emerging issues. February 18, 2020. Accessed April 9, 2022. https://www.milliman.com/-/media/milliman/pdfs/articles/2020-us-organ-tissue-transplants.ashx
- Tornai D, Szabo G. Emerging medical therapies for severe alcoholic hepatitis. Clin Mol Hepatol. 2020;26:686-696.
- Cotter TG, Sandıkçı B, Paul S, et al. Liver transplantation for alcoholic hepatitis in the United States: Excellent outcomes with profound temporal and geographic variation in frequency. Am J Transplant. 2021;21:1039-1055.
- Argemi J, Latasa MU, Atkinson SR, et al. Defective HNF4alpha-dependent gene expression as a driver of hepatocellular failure in alcoholic hepatitis. Nat Commun. 2019;10(1):3126. doi:10.1038/s41467-019-11004-3
- Wang Y, Lin W, Brown JE, et al. 25-Hydroxycholesterol 3-sulfate is an endogenous ligand of DNA methyltransferases in hepatocytes. J Lipid Res. 2021;62:100063. doi:10.1016/j.jlr.2021.100063
- Lawitz E, Hassanein T, Denham D, et al. Efficacy signals of 4-week oral DUR-928 in NASH subjects. Presented at: 2021 International Liver Conference; June 23-26, 2021; virtual. Poster no. 1198.
